Published and available for citation: Warner K, See W, Haerry D, Klingmann I, Hunter A and May M (2018) EUPATI Guidance for Patient Involvement in Medicines Research and Development (R&D); Guidance for Pharmaceutical Industry-Led Medicines R&D. Front. Med.5:270. doi: 10.3389/fmed.2018.00270
Overarching principles for patient involvement throughout the medicines research and development process
The European Patients’ Academy (EUPATI) is a pan-European Innovative Medicines Initiative (IMI) project of 33 organisations with partners from patient organisations, universities, not-for-profit organisations, and pharmaceutical companies. Throughout EUPATI the term ‘patient’ references all age groups across conditions. EUPATI does not focus on disease-specific issues or therapies, but on process of medicines development in general. Indication-specific information, age-specific or specific medicine interventions are beyond the scope of EUPATI and are the remit of health professionals as well as patient organisations. To find out more visit eupati.eu/.
The great majority of experts involved in the development and evaluation of medicines are scientists working both in the private and public sector. There is an increasing need to draw on patient knowledge and experience in order to understand what it is like to live with a specific condition, how care is administered and the day-to-day use of medicines. This input helps to improve discovery, development, and evaluation of new effective medicines.
Structured interaction with patients of all age groups and across conditions, their representatives and other stakeholders is necessary and allows the exchange of information and constructive dialogue at national and European level where the views from users of medicines can and should be considered. It is important to take into account that healthcare systems as well as practices and legislation might differ.
We recommend close cooperation and partnership between the various stakeholders including healthcare professionals’ organisations, contract research organisations, patients’ and consumers’ organisations*, academia, scientific and academic societies, regulatory authorities and health technology assessment (HTA) bodies and the pharmaceutical industry. Experience to date demonstrates that the involvement of patients has resulted in increased transparency, trust and mutual respect between them and other stakeholders.
It is acknowledged that the patients’ contribution to the discovery, development and evaluation of medicines enriches the quality of the evidence and opinion available.[1]
Existing codes of practice for patient involvement with various stakeholders do not comprehensively cover the full scope of research and development (R&D). The EUPATI guidance documents aim to support the integration of patient involvement across the entire process of medicines research and development.
EUPATI has developed these guidance documents for all stakeholders aiming to interact with patients on medicines research and development (R&D). Users may deviate from this guidance according to specific circumstances, national legislation or the unique needs of each interaction. This guidance should be adapted for individual requirements using best professional judgment.
There are four separate guidance documents covering patient involvement in:
- Pharmaceutical industry-led medicines R&D
- Ethics committees
- Regulatory authorities
- Health technology assessment (HTA).
Each guidance suggests areas where at present there are opportunities for patient involvement. This guidance should be periodically reviewed and revised to reflect evolution.
This guidance covers patient involvement in industry-led medicines R&D.
The following values are recognised in the guidance, and worked towards through the adoption of the suggested working practices (section 7). The values are:
| Relevance | Patients have knowledge, perspectives and experiences that are unique and contribute to essential evidence for industry-led research and development. |
| Fairness | Patients have the same rights to contribute to the medicines R&D process as other stakeholders and have access to knowledge and experiences that enable effective engagement. |
| Equity | Patient involvement in medicines R&D contributes to equity by seeking to understand the diverse needs of patients with particular health issues, balanced against the requirements of industry. |
| Capacity building | Patient involvement processes address barriers to involving patients in medicines R&D and build capacity of patients and research organisations to work together. |
All subsequently developed guidance should be aligned with existing national legislation covering interactions as stated in the four EUPATI guidance documents.
Disclaimer
EUPATI has developed this guidance for all stakeholders aiming to interact with patients on medicines research and development (R&D) throughout the medicines R&D lifecycle.
These guidance documents are not intended to be prescriptive and will not give detailed step-by-step advice.
This guidance should be used according to specific circumstances, national legislation or the unique needs of each interaction. This guidance should be adapted for individual requirements using best professional judgment.
Where this guidance offers advice on legal issues, it is not offered as a definitive legal interpretation and is not a substitute for formal legal advice. If formal advice is required, involved stakeholders should consult their respective legal department if available, or seek legal advice from competent sources.
EUPATI will in no event be responsible for any outcomes of any nature resulting from the use of this guidance.
The EUPATI project received support from the Innovative Medicines Initiative Joint Undertaking under grant agreement n° 115334, resources of which are composed of financial contribution from the European Union’s Seventh Framework Programme (FP7/2007-2013) and EFPIA companies.
Introduction to patient involvement in industry-led medicines R&D
The importance and merits of greater patient involvement in medicines research and development (R&D) is commonly acknowledged. A joint call for action to partner with patients in the development and lifecycle of medicines has been made by many pharmaceutical leaders[2]. The patient community likewise has called for many years for companies to embed patient involvement in medicines R&D from the earliest stages.
There is an industry-wide movement towards patient focus, with the creation of the Patient-Centered Outcomes Research Institute (PCORI), FDA’s Patient-Focused Drug Development (PFDD) initiative, Clinical Trials Transformation Initiative (CTTI) and the Patient Focused Medicine Development (PFMD) coalition. In Europe EUPATI and other IMI projects are leading the effort of generalizing patient involvement in R&D beyond specific indications. Greater patient engagement may offer many benefits for all involved parties, including the identification and understanding of unmet needs, research priorities, optimisation of clinical study design and outcome measures and endpoint development. The goal of any interaction should be to improve medicines R&D by incorporating patient needs and priorities.
The need for clear guidance on patient involvement in industry driven R&D and interaction between patients and industry is based on the following:
- Existing codes of conduct (see Appendix 1) do not thoroughly describe the involvement of patients in industry-led R&D, with exception of more general statements applicable to interaction.
- Overarching guidance on meaningful and ethical interaction is missing
- Patients and patient organisations should be involved proactively and longitudinally, especially during early discovery, development and post-approval stages of a medicine and interaction should not be confined to clinical development
- Language needs to be more directive towards patient involvement with a clear default statement that interaction is allowed unless expressly forbidden together with detailed agreement on how activities should be conducted.
- All interactions with patients should be conducted professionally, ethically and in a non-promotional manner (subject to local regulations).
Scope
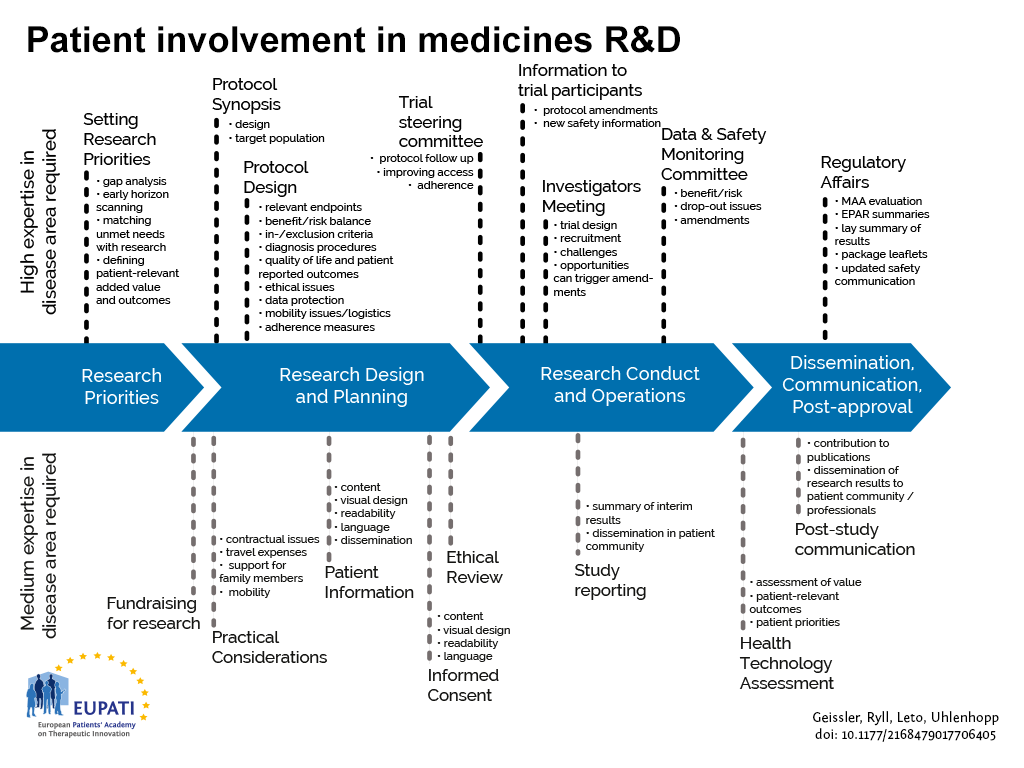
This European guidance covers the interaction between patients and the pharmaceutical industry throughout the medicines R&D lifecycle in relation to medicines for human use. This European guidance is for all functions in industry R&D on patient involvement throughout the medicines R&D lifecycle. This covers relates to activities pre-approval and post marketing, involving individuals and groups of patients. . “Patients” can be individual patients or their carers, or representatives from patient organisations with relevant expertise (section 5). See Figure 1 which indicates where patients can be involved currently; however this is not meant to limit involvement, and these opportunities may change and increase over time.
All activities should be in line with existing EU and national legislation covering pharmaceutical industry and interaction with the public. In addition, companies should follow their own internal procedures.
- Patients can be involved across the process of medicines R&D. This diagram created by Geissler, Ryll, Leto, and Uhlenhopp identifies some existing areas in which patients are involved in the process. It distinguishes between the level of expertise in a disease area that is required and the different areas where involvement can take place.
Defining “patient”
The term “patient” is often used as a general, imprecise term that does not reflect the different types of input and experience required from patients, patient advocates and patient organisations in different collaborative processes.
In order to clarify terminology for potential roles of patient interaction presented in this and the other EUPATI guidance documents, we use the term “patient” which covers the following definitions:
- “Individual Patients” are persons with personal experience of living with a disease. They may or may not have technical knowledge in R&D or regulatory processes, but their main role is to contribute with their subjective disease and treatment experience.
- “Carers” are persons supporting individual patients such as family members as well as paid or volunteer helpers.
- “Patient Advocates” are persons who have the insight and experience in supporting a larger population of patients living with a specific disease. They may or may not be affiliated with an organisation.
- “Patient Organisation Representatives” are persons who are mandated to represent and express the collective views of a patient organisation on a specific issue or disease area.
- “Patient Experts”, in addition to disease-specific expertise, have the technical knowledge in R&D and/or regulatory affairs through training or experience, for example EUPATI Fellows who have been trained by EUPATI on the full spectrum of medicines R&D.
There may be reservations about involving individual patients in collaborative activities with stakeholders on grounds that their input will be subjective and open to criticism. However, EUPATI, in line with regulatory authorities, instils the value of equity by not excluding the involvement of individuals. It should be left to the discretion of the organisation(s) initiating the interaction to choose the most adequate patient representation in terms of which type of patient for which activity (see section 7). Where an individual patient will be engaged it is suggested that the relevant patient organisation, where one exists, be informed and/or consulted to provide support and/or advice.
The type of input and mandate of the involved person should be agreed in any collaborative process prior to engagement.
Transparency
To increase transparency of patient involvement in industry-led medicines R&D, companies and patient organisations should, where allowed, publicly disclose their collaborative activities on an annual basis through their websites. Individual patient names and other protected health information should not be disclosed.
In some areas the number of experienced and knowledgeable people might be small. This fact should not prevent consultation and building on this knowledge through parallel interactions with other interested parties (such as regulatory authorities, other pharmaceutical companies) however these interactions should be disclosed.
Suggested working practices
Fostering and establishing long-term partnerships between patients, patient organisations and industry is the best approach to deliver benefits for all parties and is to be encouraged whilst respecting the independence of patients/patient organisations and other provisions set out in existing codes of conduct which would find their representation in robust, transparent operating procedures. However, it is recognised that relationship building may start with ad hoc interactions to meet short-term needs, but ideally transition to more frequent interactions as partnerships are established.
Internal cross-functional coordination in each pharmaceutical company for patient involvement would be very beneficial to all concerned, with a defined liaison role.
Pre-engagement discussions should take place to ensure mutually beneficial interaction and adequate preparation. Specific details regarding the interaction including scope, type of interaction, resource requirements and timelines should be agreed upon between patients, patient representatives and industry before interaction begins and defined in a written agreement.
Defining the interaction
Patients, patient representatives and industry should take responsibility to ensure interactions are meaningful by clearly defined processes and actions, progressed to timelines. In addition, all participants should be prepared for the interaction.
Prior to each interaction, agree mutually on (where applicable):
- the objective of the project involving patients and/or areas of common interest to establish an agreed structured interaction, providing all parties with necessary protection with regards to independence, privacy, confidentiality and expectations (see section 11, written agreement)
- the type of input and mandate of the involved person
- the tools and methods of interaction, e.g., types and frequency of meetings, ground rules, conflict resolution, evaluation
- desired patient / patient partner organisation to foster long-term working partnerships, with independence ensured (in scope)
- the profile of the type of patient/s or patient representative/s to be involved and their number
- how activity outputs will be used and ownership of outputs
- how and when the patient/s involved will be informed of outcomes
- contractual terms and conditions including consent and compensation (see section 11, written agreement).
- other elements according to the specific project
Patient identification/interaction
There are many ways to identify patients to be involved in an interaction. The main routes are through:
- existing patient organisations
- EUPATI or similar project
- advertising opportunities for patient participation
- existing relationships with healthcare providers, hospitals and researchers and other agencies
- unsolicited requests previously made by interested parties
- existing advisory boards / groups (e.g., EFPIA Think Tank, Patients and Consumers Working Party at the EMA)
- third party agencies
Compensation
It should be recognised that in many situations patients involved in activities do so voluntarily either as an individual but also when a member of an organisation. Consideration should therefore be given to:
- compensate for their total time invested plus expenses.
- any compensation offered should be fair and appropriate for the type of engagement. Ideally travel costs would be paid directly by the organising partner, rather than being reimbursed.
- covering the costs incurred by patient organizations when identifying or supporting patients for involvement in activities (i.e peer support groups, training and preparation) should also be considered.
- help organise the logistics of patient participation, including travel and/or accommodation.
Compensation also includes indirect benefits in kind (such as the a patient organisation providing services free of charge) or any other non-financial benefits in kind provided to the patient/patient organisation (such as training sessions, agency services, the setting up of web sites).
All parties should be transparent about any compensation arrangements.
Written agreement
At a minimum a written agreement should clearly define: a description of the activity and its objectives, the nature of the interaction during the activity, consent (if relevant), release, confidentiality, compensation, data privacy, compliance, declaration of conflict of interest, timelines. Interaction may only proceed on the basis of a written agreement that at a minimum spells out the basic elements of the collaboration (e.g., rules of engagement, compliance, intellectual property, financial payments).
Care should be taken so that written agreements are clear and do not limit appropriate knowledge sharing.
Events and Hospitality
The method of interaction (meetings, telephone discussions, etc.) should be discussed and mutually agreed, with convenience for patients/patient organisations as the main priority. If the interaction requires in person meetings or the development and delivery of events, these should follow existing codes of conduct, local legislation, in terms of appropriate venue/location and the level of hospitality provided.
When events are organised, the ability of any intended patient audience to attend should be considered, with appropriate measures taken to enable accessibility, assisted travel and entry into the event.
Appendix 1 – Codes of Practice Reviewed
A number of recognised codes could provide important foundation for this guidance document.
- The ECAB Protocol (description of and working procedures of ECAB (European Community Advisory Board, scientific working group at EATG, established 1997))
- Mandate, objectives and rules of procedure for the European Medicines Agency Human Scientific Committees’ Working Party with Patients' and Consumers' Organisations (PCWP) (30 May 2013)
- Minutes of EMA Human Scientific Committees’ Working Party with Patients’ and Consumers’ Organisations (PCWP) meeting with all eligible organisations (31 January 2014)
- 10 December 2009 EMA Reflection Paper on the Further Involvement of Patients and Consumers in the Agency’s Activities
- EMA leaflet on working with patients and consumers (updated 22/4/2015)
- EMA framework of interaction (revised 16 October 2014)
- Recommendations from ECAB meeting held in Bergen, Norway 1997
EATG ECAB, “The impatient Patient - From Anger to Activism”
A systematic review of the history, working models, relevance and perspectives of the European Community Advisory Board - FDA Patient Representative Program
- FDA Patient-Focused Drug Development; The Voice of the Patient: A Series of Reports from FDA's Patient-Focused Drug Development Initiative
- FDA Patient-Focused Drug Development: Enhancing Benefit-Risk Assessment in Regulatory Decision-Making
- WMA Declaration of Helsinki - Ethical Principles for Medical Research Involving Human Subjects. Retrieved 13 July, 2021, from https://www.wma.net/policies-post/wma-declaration-of-helsinki-ethical-principles-for-medical-research-involving-human-subjects/
References
- Adapted from the EMA framework. European Medicines Agency (2022). EMA/649909/2021 Adopted. https://www.ema.europa.eu/en/documents/other/engagement-framework-european-medicines-agency-and-patients-consumers-and-their-organisations_en.pdf. Last Accessed 12 February 2024.
- Hoos A, Anderson J, Boutin M, et al. 2015, Partnering with patients in the development and lifecycle of medicines: a call for action. Therapeutic Innovation & Regulatory Science.
*Consumers are recognised as stakeholders in the healthcare dialogue. The scope of EUPATI focuses on patients rather than consumers this is reflected in the educational material and guidance documents.
Webinar
A webinar took place on the 04-May-2017. You can find a video recording and the presentations used during the webinar here: https://toolbox.eupati.eu/resources/webinar-guidance-on-patient-involvement-on-industry-led-rd/
Attachments