Last update: 11 July 2023
Introduction
A Polish breast cancer patient’s organisation contacted Novartis for information on conducted clinical trials with the aim to publish the information in their site. Investigators were informed and agreed for contact with organisation. The organisation published the information making it more accessible for patients.
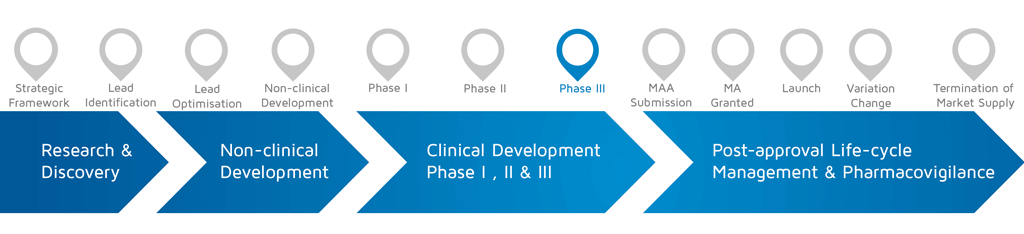
- When does it happen? – Phase III
Description of the case
Patient organisation turned to the company for information on conducted clinical trial in advanced breast cancer with the aim to publish the information on their webpage and Facebook. A prepared information sheet fitted to the requirements of social media tools and webpage (based on translated information from clinicaltrials.gov) as well as the aim to share it with the Patient Organisation was approved by the Bioethical Committee.
Novartis investigators were informed about the patient organisation request and agreed for contact with them. The patient organisation published the information using mentioned tools and making the information more accessible for patients.
Type(s) of patient (advocates) involved
- Expert patient / patient advocate with good expertise on disease, but little R&D experience.
- Patient Relations Department Oncology Business Unit from Novartis.
Benefits of patient involvement
The activity improved access to information on clinical trials for patients.
Ability to publish information on clinical trials by PAG was perceived as attempt to increase transparency in the access to information.
The interest in such information was shown by the high amount of shares (over 190) and webpage entries (over 60.000).
Challenges and barriers
Regulations regarding the access to the information on clinical trial are barriers so giving the information in the responsible way should not only be reactive but also prepared in the clear, easy to understand way and what is the most important must be approved by (Regulator’s Entity) Bioethical Committee.
Learnings
Clinicaltrials.gov is a tool out of which Patients Organisations can’t benefit because of highly complicated terminology and language barrier.
There is a need to educate and build awareness with the general public about clinical trials.
When the external entity approval process is included in the activity it impacts the timing of activity.
A3-patients-organisations-access-to-information-V1.0