Discovery and development of medicines
By studying a disease, scientists can understand what causes the illness and which cells and their receptors are involved. This research can help scientists to develop new medicines to treat diseases and to relieve symptoms.

It takes well over 10 years of careful planning and research for a medicine to go from molecule to a marketable treatment.
From discovery of molecules to development of medicine
Once a target receptor molecule or an enzyme has been identified, scientists begin to look for potential compounds that will interact with the target to correct disease-related activity. This research can involve searching through ‘libraries’ of millions of molecules that have been developed by pharmaceutical companies. Large-scale testing of potentially useful molecules from the ‘libraries’ (also called high-throughput screening), is conducted to discover which ones will bind to the target receptor.
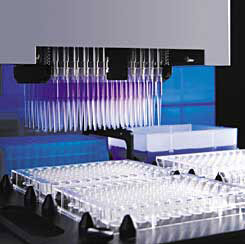
Part of the high-throughput screening process. A machine with many pipettes and vials allows the simultaneous testing a large amount of potentially useful molecules.
The most promising molecules discovered will then be modified in many ways to try to make an effective medicine that has few negative side effects. When scientists find a molecule with the desired properties of a potential medicine, testing in animals is often necessary. If this testing shows the medicine is safe, the process will continue to testing in humans. Finally, regulatory authorities will consider whether or not to approve the medicine for use in humans.
The ideal molecule should:
- reach the right part of the body to hit the target (ideally only the target and no other healthy cells or enzymes)
- have few side effects
- be absorbed by the body
- remain in the body long enough to have the desired effect
- be able to be manufactured in sufficient quantities
- have a long shelf-life as a medicine.
Attachments
- Presentation: Discovery and Development of Medicines
Size: 862,301 bytes, Format: .pptx
A presentation describing the discovery and development of medicines, which can be adapted for own use.
A2-1.06.3-V1.2